Element's Name: Potassium
Chemical Symbol: K
Atomic Number: 19
Average Atomic Mass: 39.0983 ± 0.0001 u
Chemical Symbol: K
Atomic Number: 19
Average Atomic Mass: 39.0983 ± 0.0001 u
Potassium's Chemical Symbol is K, it's atomic number is 19. It's average atomic mass is 39.10, while Potassium's density is 0.89 g/cm3. The location that this element can be found in is group 1, period 4. Elemental potassium is a soft silvery-white alkali metal. It oxidizes rapidly in air and explodes when in water.
Humphry Davy is the one who discovered potassium. He was a Cornish chemist and inventor. The word Potassium comes from the English word potash, and it's chemical symbol comes from the latin word for alkali, kalium. Potassium is a solid at average room temperature, but above 63.3 celsius, it is a liquid. Also, above 760 celsius, potassium becomes a gas.
Potassium melts at 63.65 celsius. Potassium can not freeze, as it is already a solid at room temperature. The boiling point of Potassium is 774.0 celsius.
Some uses of potassium are quite everyday in our lives. Of course you know that potassium is in bananas. Potassium can also be found in the ground, and most soil, also used as a nutrient in fertilizer. Also, it is used in baking powder and is a good food preservative. Potassium has it's fun sides too, as it is also used in fireworks, explosives, safety matches, tanning of leather, and fly paper.
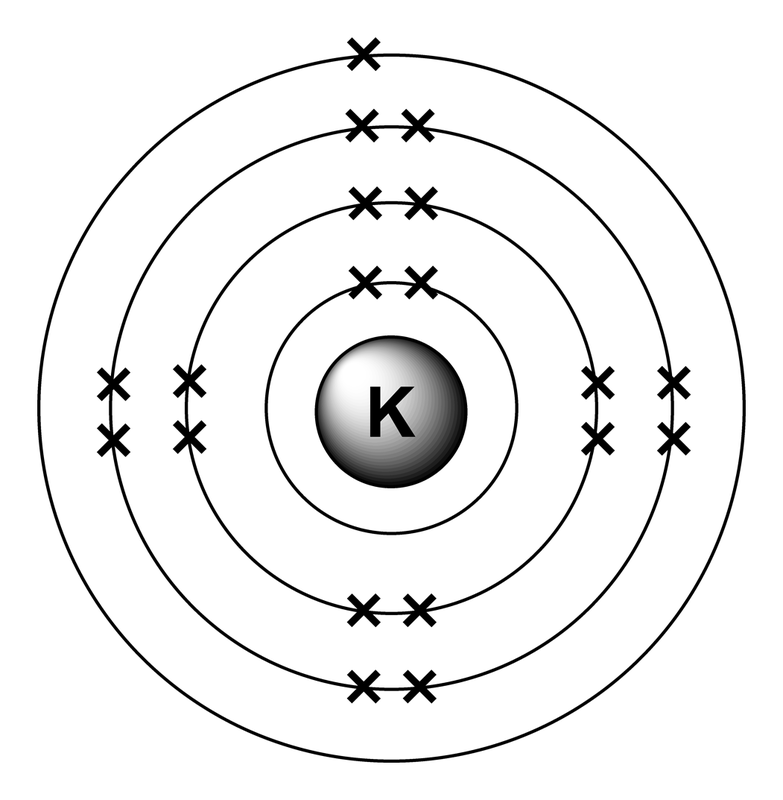
Potassium has 0 electrons, but has one valence electron in it's outer shell, but when it gives away that electron, it now becomes Potassium ion.
Humphry Davy is the one who discovered potassium. He was a Cornish chemist and inventor. The word Potassium comes from the English word potash, and it's chemical symbol comes from the latin word for alkali, kalium. Potassium is a solid at average room temperature, but above 63.3 celsius, it is a liquid. Also, above 760 celsius, potassium becomes a gas.
Potassium melts at 63.65 celsius. Potassium can not freeze, as it is already a solid at room temperature. The boiling point of Potassium is 774.0 celsius.
Some uses of potassium are quite everyday in our lives. Of course you know that potassium is in bananas. Potassium can also be found in the ground, and most soil, also used as a nutrient in fertilizer. Also, it is used in baking powder and is a good food preservative. Potassium has it's fun sides too, as it is also used in fireworks, explosives, safety matches, tanning of leather, and fly paper.
Potassium has 0 electrons, but has one valence electron in it's outer shell, but when it gives away that electron, it now becomes Potassium ion.
Elemental Potassium reacting to water. ^^
The electron configuration of Potassium. ^^
Credits:
http://antoine.frostburg.edu/chem/senese/101/history/faq/sodium-discovery.shtmlhttp://www.ptable.com
http://antoine.frostburg.edu/chem/senese/101/history/faq/sodium-discovery.shtmlhttp://www.ptable.com